 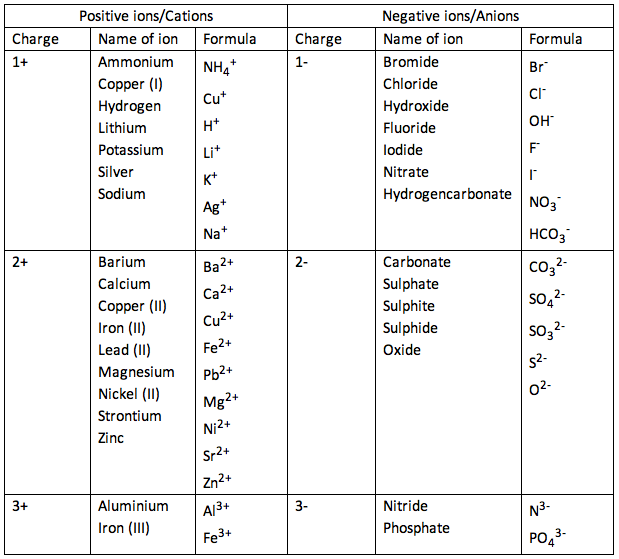
For hydrogen, there is only one electron and so the nuclear charge ( Z) and the effective nuclear charge ( Z eff) are equal. This is the pull exerted on a specific electron by the nucleus, taking into account any electron–electron repulsions. Please re-write this article to improve it to explain concepts before they are used in answers or taken for granted as prior knowledge when they have not first been defined in Unit 1.\). The problems are then for ionic compounds that are not monoatomic. What is the trend in the ionic charges of the elements in Groups 1, 2, and 13 of the periodic table (b) What is the trend in ionic charges of the elements. The use of magnitude is used without being introduced followed by a statement that isn't well described, 'The magnitude of the charge is equal to the number of electrons lost, which is equal to the number of valence electrons in the neutral atom.'Īluminum is in group 13, or IIIA is stated when no introduction has been provided to what IIIA means. Where in Unit 1 is s, p, d, f and shell and subshell defined prior to this article. 
The alkali metals (group 1) have ns1 valence electron configurations and form M + ions, while the alkaline earth metals (group 2) have ns2 valence electron configurations and form M 2+ ions. Group 2A (or IIA) of the periodic table are the alkaline earth metals: beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium. Only carbon has an atomic mass value that is an integer. IUPAC group number is also not described, it is simply used, without definition or prior description.Įlectron configuration is used as part of a hidden explanation as an answer, yet the article or Unit 1 does not explain what an electron configuration is. The chemical families consist of elements that have the same valence electron configuration and tend to have similar chemistry. An amu is 1/12 the mass of one atom of 12C carbon or about 1.66 x 10-27 kg. The first example occurs in the case of the lanthanoids (elements having atomic numbers between 57 and 71).The lanthanoids have the general electron configuration Kr4d 10 4f i 5s 2 5p 6 5d 0 or 1 6s 2. As early chemists discovered more elements, they realized that. Because the group 13 elements generally contain only six valence electrons in their neutral compounds, these compounds are all moderately strong Lewis acids. The metallic elements in groups other than 1, 2, or 3 also lose. The third major category of elements arises when the distinguishing electron occupies an f subshell. Periodic Table With Common Ionic Charges Use the periodic table to predict oxidation state Todd Helmenstine By Anne Marie Helmenstine, Ph.D. The heavier elements in the group can also form compounds in the +1 oxidation state formed by the formal loss of the single np valence electron. Where in Unit 1 is Octet and Valence given definition. Image of a periodic table showing the charges on the monatomic cations: +1 for. Octet is used without definition, followed by valence also being used without definition. 
We are being asked to 'Remember that periodic groups refer to columns on the periodic table, whereas rows are known as periods' when this concept has also not been initially defined in this article or in Unit 1. Where is monatomic defined in this article or in Unit 1. Group number Number of valence electrons. Words are used without first having had a definition or explanation in the article or earlier in Unit 1. Thus, members of a family in the periodic table share some properties. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
